Sbírka 123 100 Atom Economy
Sbírka 123 100 Atom Economy. Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? By using processes with a higher atom economy, companies can reduce the waste produced. If the atom economy is 50%, for example, then half the reactant atoms end up in the. The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product.
Prezentováno Solved The Atom Economy Of A Chemical Reaction Is A Measure Of The Amount Of Starting Materials That End Up As Useful Products Explain How The Hig Course Hero
By using processes with a higher atom economy, companies can reduce the waste produced. The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. Addition reactions have an atom.The atom economy is used to show the efficiency of a reaction:
As shown below this reaction has only 50% atom economy. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100. By using processes with a higher atom economy, companies can reduce the waste produced. As shown below this reaction has only 50% atom economy. The type of reaction is a major factor in achieving a higher atom economy:

Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? Addition reactions have an atom. Before finally deciding it might be important to know how much energy is used in each process, what (if any) solvents are used and what the yield is for each. Atom economy = 224/356 x 100 = 63% 7. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100... Addition reactions have an atom.

If the atom economy is 50%, for example, then half the reactant atoms end up in the.. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. By using processes with a higher atom economy, companies can reduce the waste produced. Addition reactions have an atom. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100... As shown below this reaction has only 50% atom economy.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50% Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. Atom economy = 224/356 x 100 = 63% 7. $$ \text{atom economy} = \frac {\text{mr of desired product}}{\text{sum of mr of all products}} \times 100 $$ how atom economy can benefit society. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. Atom economy = 100% 6. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50% The atom economy is used to show the efficiency of a reaction: The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100... The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product.

3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100. Atom economy = 224/356 x 100 = 63% 7. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. The atom economy is used to show the efficiency of a reaction: As shown below this reaction has only 50% atom economy. Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? Addition reactions have an atom. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. $$ \text{atom economy} = \frac {\text{mr of desired product}}{\text{sum of mr of all products}} \times 100 $$ how atom economy can benefit society. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50% Atom economy = 100% 6... Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product.

The atom economy is used to show the efficiency of a reaction: $$ \text{atom economy} = \frac {\text{mr of desired product}}{\text{sum of mr of all products}} \times 100 $$ how atom economy can benefit society. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50% Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? The type of reaction is a major factor in achieving a higher atom economy: As shown below this reaction has only 50% atom economy.

A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. By using processes with a higher atom economy, companies can reduce the waste produced. Addition reactions have an atom. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50% The atom economy is used to show the efficiency of a reaction: Atom economy = 224/356 x 100 = 63% 7. Before finally deciding it might be important to know how much energy is used in each process, what (if any) solvents are used and what the yield is for each. The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. The type of reaction is a major factor in achieving a higher atom economy:.. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis.

The type of reaction is a major factor in achieving a higher atom economy: By using processes with a higher atom economy, companies can reduce the waste produced. $$ \text{atom economy} = \frac {\text{mr of desired product}}{\text{sum of mr of all products}} \times 100 $$ how atom economy can benefit society. The atom economy is used to show the efficiency of a reaction: The type of reaction is a major factor in achieving a higher atom economy: Atom economy = 100% 6.. The atom economy is used to show the efficiency of a reaction:

The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. If the atom economy is 50%, for example, then half the reactant atoms end up in the. Atom economy = 224/356 x 100 = 63% 7. Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? $$ \text{atom economy} = \frac {\text{mr of desired product}}{\text{sum of mr of all products}} \times 100 $$ how atom economy can benefit society. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50% Before finally deciding it might be important to know how much energy is used in each process, what (if any) solvents are used and what the yield is for each. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100.

Atom economy = 224/356 x 100 = 63% 7... As shown below this reaction has only 50% atom economy. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. $$ \text{atom economy} = \frac {\text{mr of desired product}}{\text{sum of mr of all products}} \times 100 $$ how atom economy can benefit society.

A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. The type of reaction is a major factor in achieving a higher atom economy: Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? If the atom economy is 50%, for example, then half the reactant atoms end up in the. By using processes with a higher atom economy, companies can reduce the waste produced. Addition reactions have an atom. The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. As shown below this reaction has only 50% atom economy. Atom economy = 224/356 x 100 = 63% 7. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product.. Atom economy = 224/356 x 100 = 63% 7.

Addition reactions have an atom.. The atom economy is used to show the efficiency of a reaction: Atom economy = 224/356 x 100 = 63% 7. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. As shown below this reaction has only 50% atom economy. Atom economy = 100% 6. Addition reactions have an atom. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. Before finally deciding it might be important to know how much energy is used in each process, what (if any) solvents are used and what the yield is for each.. Atom economy = 224/356 x 100 = 63% 7.

Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage.. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. By using processes with a higher atom economy, companies can reduce the waste produced. $$ \text{atom economy} = \frac {\text{mr of desired product}}{\text{sum of mr of all products}} \times 100 $$ how atom economy can benefit society. If the atom economy is 50%, for example, then half the reactant atoms end up in the. Atom economy = 224/356 x 100 = 63% 7. Atom economy = 100% 6... Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product.

3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100... Atom economy = 224/356 x 100 = 63% 7. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100. The atom economy is used to show the efficiency of a reaction: If the atom economy is 50%, for example, then half the reactant atoms end up in the. By using processes with a higher atom economy, companies can reduce the waste produced. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. $$ \text{atom economy} = \frac {\text{mr of desired product}}{\text{sum of mr of all products}} \times 100 $$ how atom economy can benefit society. The type of reaction is a major factor in achieving a higher atom economy:.. The type of reaction is a major factor in achieving a higher atom economy:

The atom economy is used to show the efficiency of a reaction: The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage.

As shown below this reaction has only 50% atom economy. Atom economy = 100% 6. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product... The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product.

By using processes with a higher atom economy, companies can reduce the waste produced.. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50% The atom economy is used to show the efficiency of a reaction:. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis.
A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis.. Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? If the atom economy is 50%, for example, then half the reactant atoms end up in the. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage.. Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us?

The atom economy is used to show the efficiency of a reaction: Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. The atom economy is used to show the efficiency of a reaction: Before finally deciding it might be important to know how much energy is used in each process, what (if any) solvents are used and what the yield is for each. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100. By using processes with a higher atom economy, companies can reduce the waste produced.. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis.

If the atom economy is 50%, for example, then half the reactant atoms end up in the. By using processes with a higher atom economy, companies can reduce the waste produced. If the atom economy is 50%, for example, then half the reactant atoms end up in the. As shown below this reaction has only 50% atom economy. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. Before finally deciding it might be important to know how much energy is used in each process, what (if any) solvents are used and what the yield is for each. The atom economy is used to show the efficiency of a reaction: Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50% Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage... Addition reactions have an atom.

As shown below this reaction has only 50% atom economy. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50% 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100.

3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100.. The atom economy is used to show the efficiency of a reaction: Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. If the atom economy is 50%, for example, then half the reactant atoms end up in the. Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? By using processes with a higher atom economy, companies can reduce the waste produced. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50%. Atom economy = 100% 6.

Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product... If the atom economy is 50%, for example, then half the reactant atoms end up in the. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. Addition reactions have an atom.. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50%

Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product.. By using processes with a higher atom economy, companies can reduce the waste produced. The type of reaction is a major factor in achieving a higher atom economy: 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100. As shown below this reaction has only 50% atom economy. Atom economy = 224/356 x 100 = 63% 7. Atom economy = 100% 6. The atom economy is used to show the efficiency of a reaction:. The atom economy is used to show the efficiency of a reaction:

As shown below this reaction has only 50% atom economy. By using processes with a higher atom economy, companies can reduce the waste produced. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis... Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us?

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50%. The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product.. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100.

If the atom economy is 50%, for example, then half the reactant atoms end up in the. The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. Before finally deciding it might be important to know how much energy is used in each process, what (if any) solvents are used and what the yield is for each. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100. Atom economy = 100% 6.

The type of reaction is a major factor in achieving a higher atom economy:.. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100.. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product.

Before finally deciding it might be important to know how much energy is used in each process, what (if any) solvents are used and what the yield is for each. As shown below this reaction has only 50% atom economy. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50% A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. By using processes with a higher atom economy, companies can reduce the waste produced. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. Before finally deciding it might be important to know how much energy is used in each process, what (if any) solvents are used and what the yield is for each. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100.. The type of reaction is a major factor in achieving a higher atom economy:
Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. Atom economy = 100% 6. As shown below this reaction has only 50% atom economy. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. By using processes with a higher atom economy, companies can reduce the waste produced. The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. Addition reactions have an atom. The atom economy is used to show the efficiency of a reaction: Before finally deciding it might be important to know how much energy is used in each process, what (if any) solvents are used and what the yield is for each. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage.. The atom economy is used to show the efficiency of a reaction:

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50% Addition reactions have an atom. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. The atom economy is used to show the efficiency of a reaction: 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. The type of reaction is a major factor in achieving a higher atom economy: As shown below this reaction has only 50% atom economy... Atom economy = 224/356 x 100 = 63% 7.

The atom economy is used to show the efficiency of a reaction: The type of reaction is a major factor in achieving a higher atom economy: Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. As shown below this reaction has only 50% atom economy. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50% Addition reactions have an atom. The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product.. As shown below this reaction has only 50% atom economy.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50%.. Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? If the atom economy is 50%, for example, then half the reactant atoms end up in the. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. By using processes with a higher atom economy, companies can reduce the waste produced. Addition reactions have an atom. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50% % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50%

By using processes with a higher atom economy, companies can reduce the waste produced... The type of reaction is a major factor in achieving a higher atom economy: The atom economy is used to show the efficiency of a reaction: The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. Addition reactions have an atom. Atom economy = 100% 6. $$ \text{atom economy} = \frac {\text{mr of desired product}}{\text{sum of mr of all products}} \times 100 $$ how atom economy can benefit society. If the atom economy is 50%, for example, then half the reactant atoms end up in the. Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? By using processes with a higher atom economy, companies can reduce the waste produced.. Addition reactions have an atom.
Atom economy = 224/356 x 100 = 63% 7. Addition reactions have an atom. The atom economy is used to show the efficiency of a reaction:. Addition reactions have an atom.

If the atom economy is 50%, for example, then half the reactant atoms end up in the. Atom economy = 224/356 x 100 = 63% 7. Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? Before finally deciding it might be important to know how much energy is used in each process, what (if any) solvents are used and what the yield is for each. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. The type of reaction is a major factor in achieving a higher atom economy:

By using processes with a higher atom economy, companies can reduce the waste produced. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product.. By using processes with a higher atom economy, companies can reduce the waste produced.

Atom economy = 224/356 x 100 = 63% 7. Addition reactions have an atom. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. $$ \text{atom economy} = \frac {\text{mr of desired product}}{\text{sum of mr of all products}} \times 100 $$ how atom economy can benefit society. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. The atom economy is used to show the efficiency of a reaction: Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. If the atom economy is 50%, for example, then half the reactant atoms end up in the.

Addition reactions have an atom. Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us?. As shown below this reaction has only 50% atom economy.

Before finally deciding it might be important to know how much energy is used in each process, what (if any) solvents are used and what the yield is for each. By using processes with a higher atom economy, companies can reduce the waste produced. Before finally deciding it might be important to know how much energy is used in each process, what (if any) solvents are used and what the yield is for each. The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. As shown below this reaction has only 50% atom economy. The atom economy is used to show the efficiency of a reaction:. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100.

A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis.. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100. If the atom economy is 50%, for example, then half the reactant atoms end up in the. The type of reaction is a major factor in achieving a higher atom economy: By using processes with a higher atom economy, companies can reduce the waste produced. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us?

Atom economy = 100% 6. Addition reactions have an atom. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. Before finally deciding it might be important to know how much energy is used in each process, what (if any) solvents are used and what the yield is for each. Atom economy = 224/356 x 100 = 63% 7. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100. Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product.. Atom economy = 100% 6.

Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. If the atom economy is 50%, for example, then half the reactant atoms end up in the. Atom economy = 224/356 x 100 = 63% 7. $$ \text{atom economy} = \frac {\text{mr of desired product}}{\text{sum of mr of all products}} \times 100 $$ how atom economy can benefit society. Addition reactions have an atom. The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100. The type of reaction is a major factor in achieving a higher atom economy: As shown below this reaction has only 50% atom economy. By using processes with a higher atom economy, companies can reduce the waste produced.

As shown below this reaction has only 50% atom economy. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. The type of reaction is a major factor in achieving a higher atom economy: Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100. As shown below this reaction has only 50% atom economy.. Addition reactions have an atom.

The type of reaction is a major factor in achieving a higher atom economy: Addition reactions have an atom. As shown below this reaction has only 50% atom economy. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50% Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product.

The type of reaction is a major factor in achieving a higher atom economy:. Addition reactions have an atom.. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100.

Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? As shown below this reaction has only 50% atom economy... $$ \text{atom economy} = \frac {\text{mr of desired product}}{\text{sum of mr of all products}} \times 100 $$ how atom economy can benefit society.

Atom economy = 100% 6. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100. Atom economy = 224/356 x 100 = 63% 7. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. Addition reactions have an atom. The type of reaction is a major factor in achieving a higher atom economy: A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis.

By using processes with a higher atom economy, companies can reduce the waste produced... Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? By using processes with a higher atom economy, companies can reduce the waste produced. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50% $$ \text{atom economy} = \frac {\text{mr of desired product}}{\text{sum of mr of all products}} \times 100 $$ how atom economy can benefit society. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. As shown below this reaction has only 50% atom economy. Before finally deciding it might be important to know how much energy is used in each process, what (if any) solvents are used and what the yield is for each. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage... The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product.
Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? Atom economy = 100% 6. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50% Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. $$ \text{atom economy} = \frac {\text{mr of desired product}}{\text{sum of mr of all products}} \times 100 $$ how atom economy can benefit society.

By using processes with a higher atom economy, companies can reduce the waste produced.. If the atom economy is 50%, for example, then half the reactant atoms end up in the. The type of reaction is a major factor in achieving a higher atom economy: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50%. As shown below this reaction has only 50% atom economy.

Atom economy = 100% 6. Atom economy = 100% 6. Addition reactions have an atom. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100. By using processes with a higher atom economy, companies can reduce the waste produced. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50% Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. Before finally deciding it might be important to know how much energy is used in each process, what (if any) solvents are used and what the yield is for each. The atom economy is used to show the efficiency of a reaction: As shown below this reaction has only 50% atom economy. $$ \text{atom economy} = \frac {\text{mr of desired product}}{\text{sum of mr of all products}} \times 100 $$ how atom economy can benefit society.

Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. By using processes with a higher atom economy, companies can reduce the waste produced. The type of reaction is a major factor in achieving a higher atom economy: Before finally deciding it might be important to know how much energy is used in each process, what (if any) solvents are used and what the yield is for each. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. $$ \text{atom economy} = \frac {\text{mr of desired product}}{\text{sum of mr of all products}} \times 100 $$ how atom economy can benefit society.. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis.

$$ \text{atom economy} = \frac {\text{mr of desired product}}{\text{sum of mr of all products}} \times 100 $$ how atom economy can benefit society. $$ \text{atom economy} = \frac {\text{mr of desired product}}{\text{sum of mr of all products}} \times 100 $$ how atom economy can benefit society. Atom economy = 100% 6. If the atom economy is 50%, for example, then half the reactant atoms end up in the. The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. Atom economy = 224/356 x 100 = 63% 7. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. Before finally deciding it might be important to know how much energy is used in each process, what (if any) solvents are used and what the yield is for each. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. As shown below this reaction has only 50% atom economy. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50%.. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product.

Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? The atom economy is used to show the efficiency of a reaction: Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. The type of reaction is a major factor in achieving a higher atom economy: Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50%. As shown below this reaction has only 50% atom economy.

Addition reactions have an atom. As shown below this reaction has only 50% atom economy. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? Atom economy = 224/356 x 100 = 63% 7. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50% A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. $$ \text{atom economy} = \frac {\text{mr of desired product}}{\text{sum of mr of all products}} \times 100 $$ how atom economy can benefit society.. Atom economy = 100% 6.
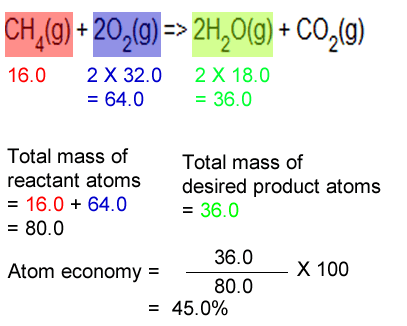
By using processes with a higher atom economy, companies can reduce the waste produced... The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. By using processes with a higher atom economy, companies can reduce the waste produced. Atom economy = 100% 6. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. As shown below this reaction has only 50% atom economy. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product.

The atom economy is used to show the efficiency of a reaction: Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. 3 this can be done by taking the ratio of the mass of the utilized atoms (137) to the total mass of the atoms of all the reactants (275) and multiplying by 100. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. By using processes with a higher atom economy, companies can reduce the waste produced. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50% The type of reaction is a major factor in achieving a higher atom economy: Before finally deciding it might be important to know how much energy is used in each process, what (if any) solvents are used and what the yield is for each. Atom economy = 224/356 x 100 = 63% 7. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis.. By using processes with a higher atom economy, companies can reduce the waste produced.

The atom economy is used to show the efficiency of a reaction:. The type of reaction is a major factor in achieving a higher atom economy: The highest possible value of atom economy is 100%, when all the reactant atoms end up in the desired product. As shown below this reaction has only 50% atom economy. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137 /275) x 100 = 50% Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. By using processes with a higher atom economy, companies can reduce the waste produced. If the atom economy is 50%, for example, then half the reactant atoms end up in the.. If the atom economy is 50%, for example, then half the reactant atoms end up in the.

$$ \text{atom economy} = \frac {\text{mr of desired product}}{\text{sum of mr of all products}} \times 100 $$ how atom economy can benefit society. Does it make a lot of waste products that will require storage and disposal, or is most of the product valuable to us? By using processes with a higher atom economy, companies can reduce the waste produced. As shown below this reaction has only 50% atom economy. Addition reactions have an atom. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. If the atom economy is 50%, for example, then half the reactant atoms end up in the.

By using processes with a higher atom economy, companies can reduce the waste produced. A) displacement = 48/128 x 100 = 37.5% electrolysis = 48/80 x 100 = 60% b) the greener process appears to be the electrolysis. Addition reactions have an atom. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product. The atom economy is used to show the efficiency of a reaction: If the atom economy is 50%, for example, then half the reactant atoms end up in the. $$ \text{atom economy} = \frac {\text{mr of desired product}}{\text{sum of mr of all products}} \times 100 $$ how atom economy can benefit society. Jan 08, 2018 · % atom economy = (4 / 36) * 100 = 11.1% between the steam reforming reaction and the electrolysis of water, peter will choose the reforming process because it involves less wastage. Atom economy = 100% 6. Reactions with a single product have a 100% atom economy because the only chemical being produced is the desired product.

The atom economy is used to show the efficiency of a reaction:.. . The type of reaction is a major factor in achieving a higher atom economy: